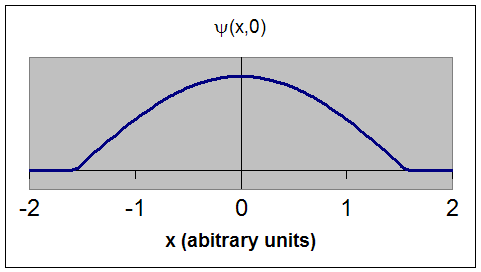
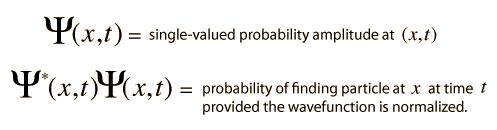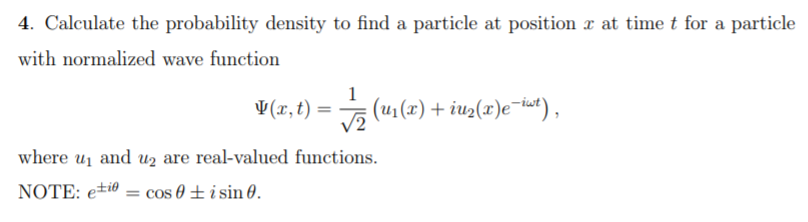Physics - Ch 66 Ch 4 Quantum Mechanics: Schrodinger Eqn (21 of 72) Prob. of Finding Particle 1 - YouTube
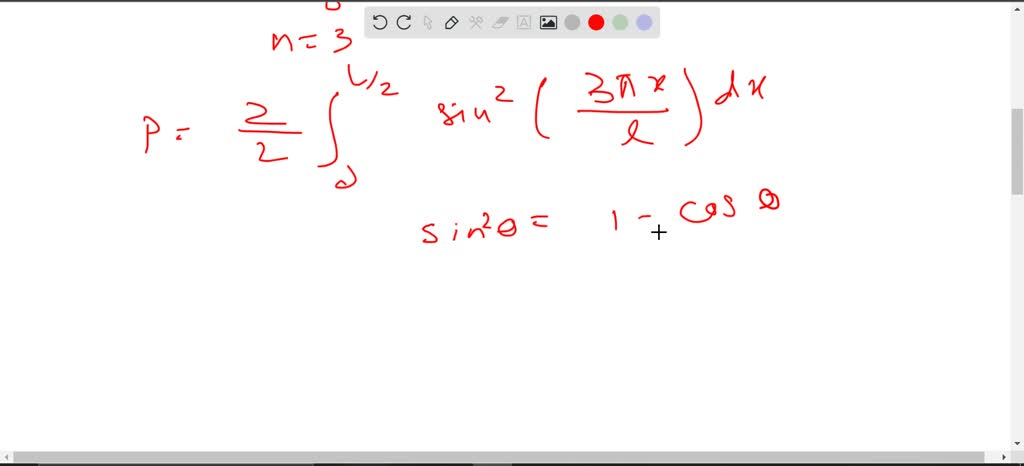
SOLVED: Consider the wave function for a particle in a one-dimensional box when the level is n = 3. Calculate the total probability of finding the particle between x = 0 and

Physics - Ch 66 Ch 4 Quantum Mechanics: Schrodinger Eqn (26 of 92) Prob. of a Particle 1-D Box n=2 - YouTube

SOLVED: A particle confined within a one-dimensional region 0 < x < a can be described by the wave function w(x,t) = Asin ict (a) Find the potential energy V(x). Find the

Does the probability of finding a particle in the marked region depend... | Download Scientific Diagram
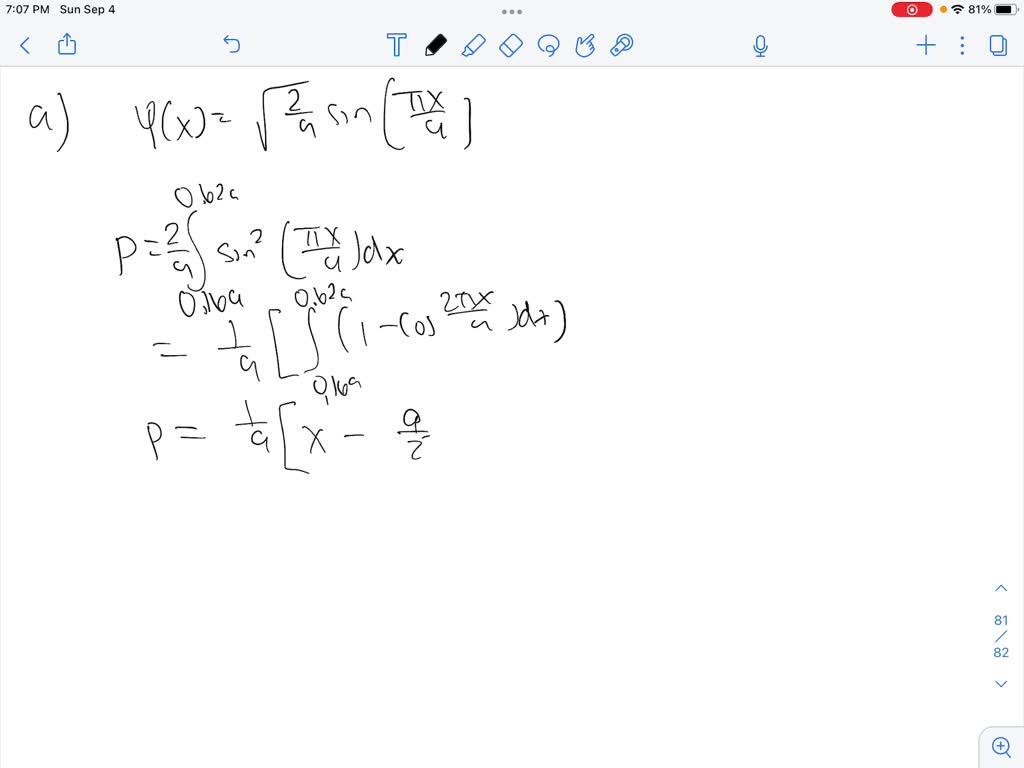
SOLVED: If a particle has wave function , the probability of finding the particle between any two points and is: (a) For a particle in the ground state of a rigid box,
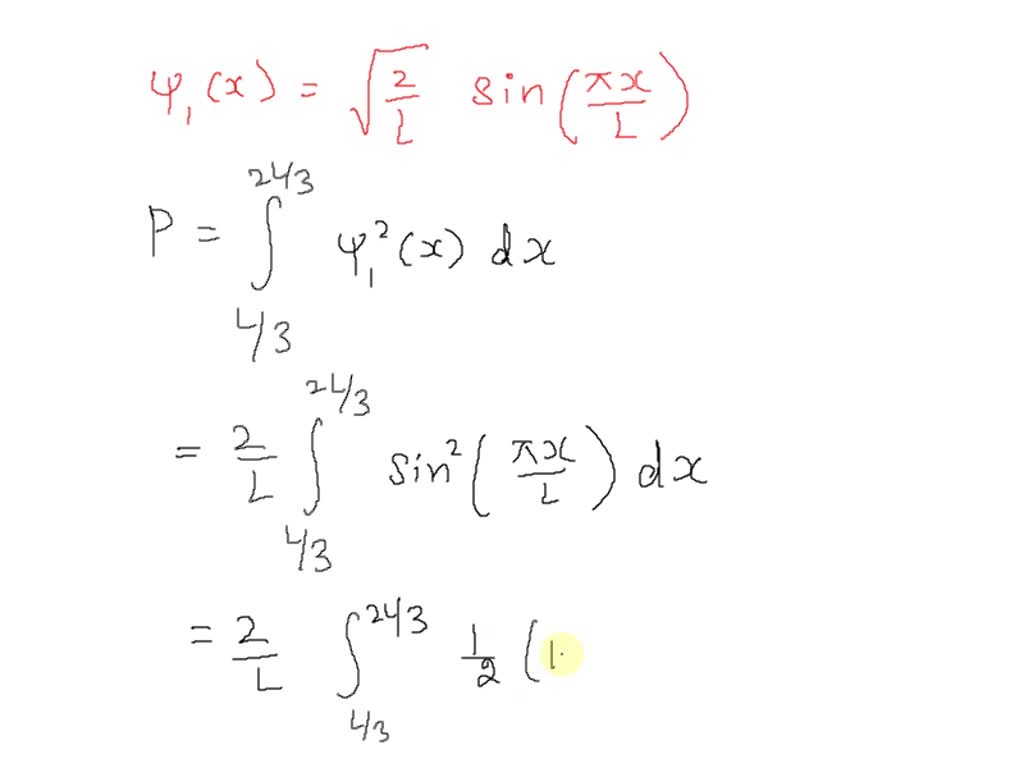
SOLVED: 3. Derive and expression for the probability of finding particle in the first quarter of a one-dimensional box of length a. Your expression should be as a function of the quantum

Find the probability that a particle in a box L wide can be nd between x - nd x - L n when it is - YouTube

quantum mechanics - Differences between wavefunction, probability and probability density? - Physics Stack Exchange

Physics - Ch 66 Ch 4 Quantum Mechanics: Schrodinger Eqn (26 of 92) Prob. of a Particle 1-D Box n=2 - YouTube
What is the probability of finding a particle in a one-dimensional box in energy level n=4 between x=L/4 and x=L/2? L is the length of the box. - Quora








![Solved 2. 160 points] Consider a particle of massmo in a | Chegg.com Solved 2. 160 points] Consider a particle of massmo in a | Chegg.com](https://media.cheggcdn.com/media%2F864%2F8647c20e-28a6-4704-abc2-d900ec891f2a%2Fimage)