Recent developments on the epoxidation of alkenes using hydrogen peroxide as an oxidant - Green Chemistry (RSC Publishing) DOI:10.1039/B208925B
Vanadium-doped phosphomolybdic acids as catalysts for geraniol oxidation with hydrogen peroxide - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01258H

PDF) Opposite regioselectivity in the epoxidation of geraniol and linalool with molybdenum and tungsten peroxo complexes | Gaetano Tomaselli - Academia.edu

Mechanistic Insights on Skin Sensitization to Linalool Hydroperoxides: EPR Evidence on Radical Intermediates Formation in Reconstructed Human Epidermis and 13C NMR Reactivity Studies with Thiol Residues | Chemical Research in Toxicology
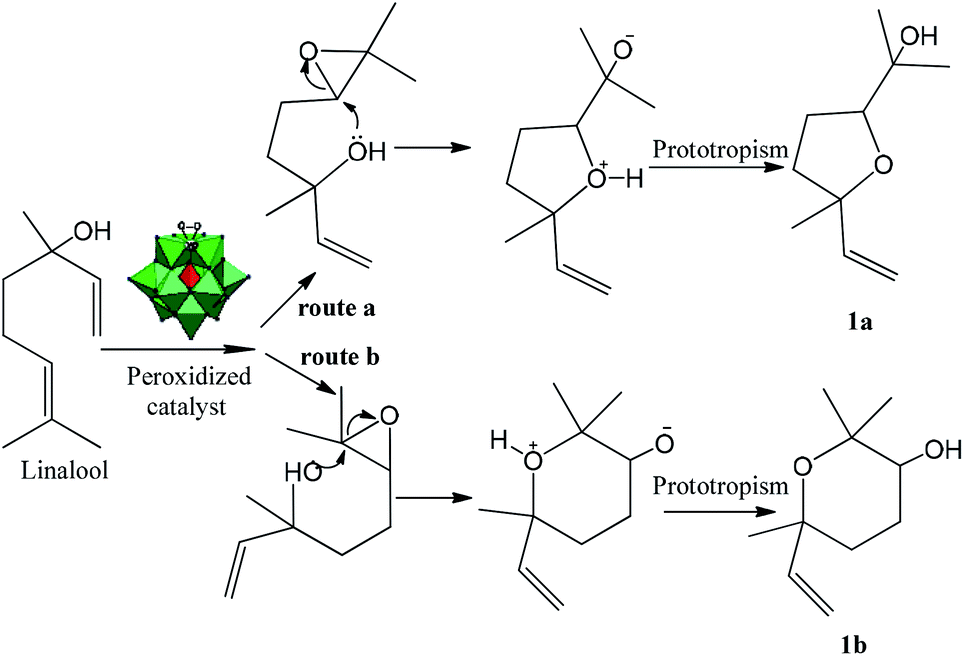
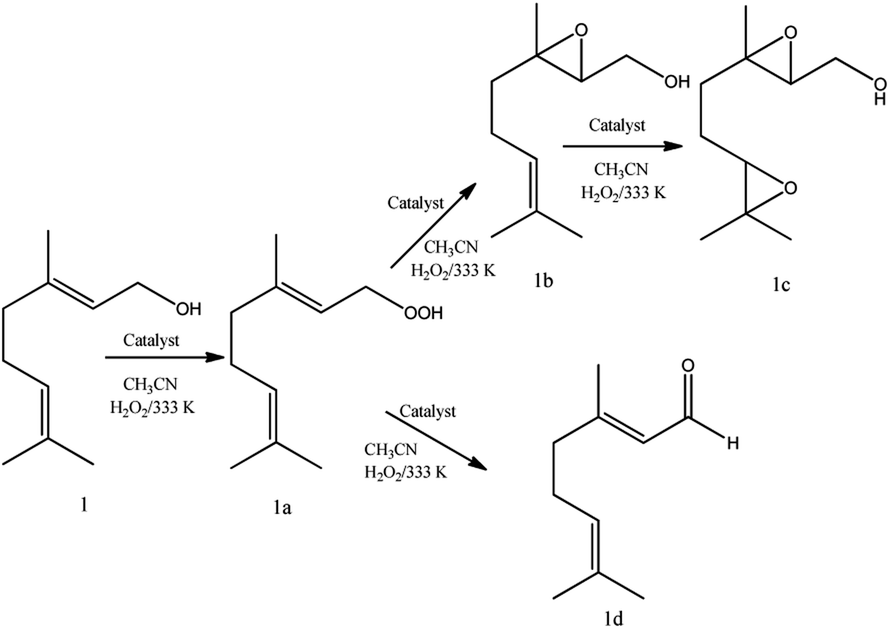
One-pot synthesis at room temperature of epoxides and linalool derivative pyrans in monolacunary Na 7 PW 11 O 39 -catalyzed oxidation reactions by hyd ... - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00047G

Ultrasonic and Catalyst-Free Epoxidation of Limonene and Other Terpenes Using Dimethyl Dioxirane in Semibatch Conditions | ACS Sustainable Chemistry & Engineering

PDF) One-pot synthesis at room temperature of epoxides and linalool derivative pyrans in monolacunary Na 7 PW 11 O 39 -catalyzed oxidation reactions by hydrogen peroxide †

ChemInform Abstract: Effect of Additives on Chemoselectivity and Diastereoselectivity in the Catalytic Epoxidation of Chiral Allylic Alcohols with Hydrogen Peroxide and Binuclear Manganese Complexes. | Request PDF
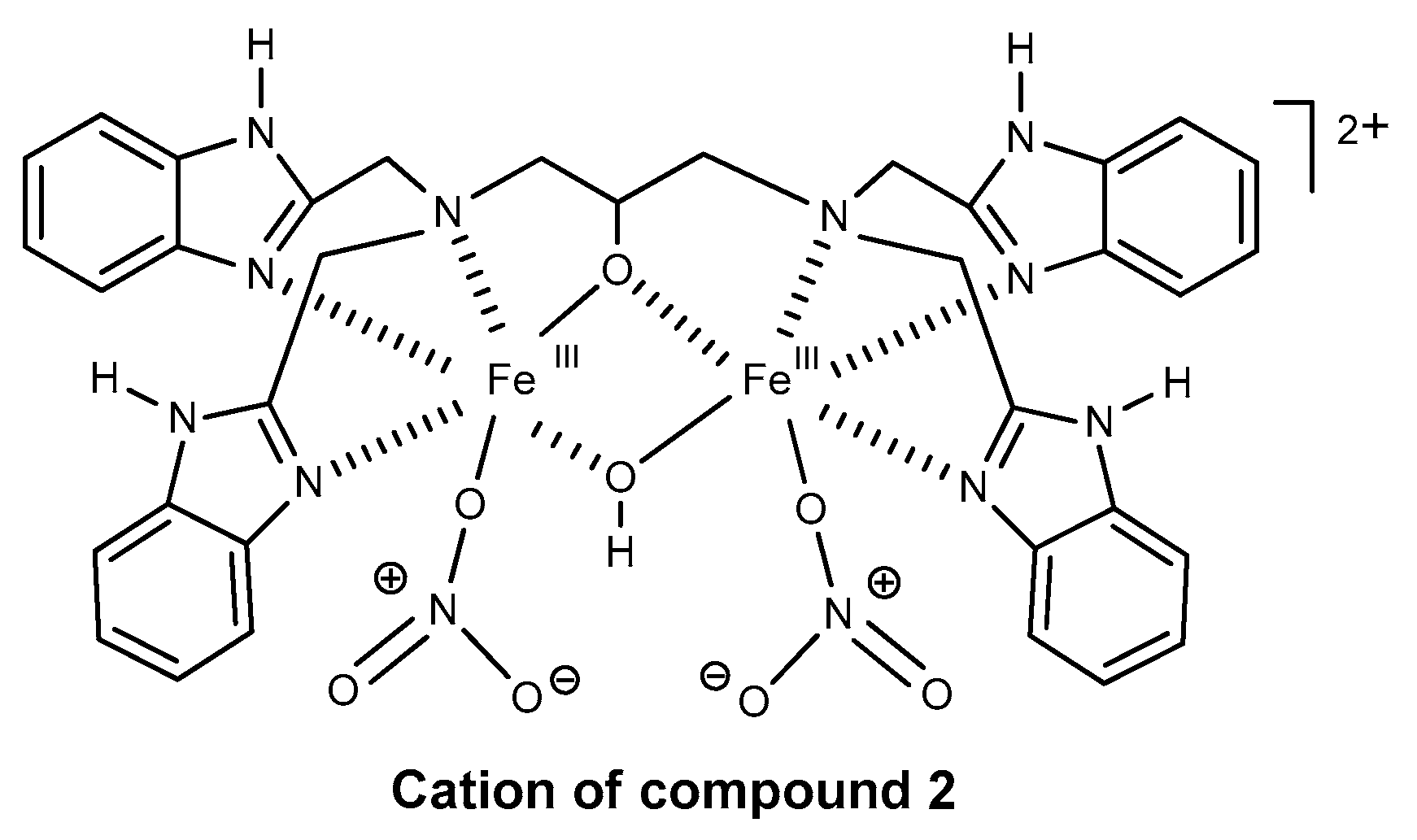
Catalysts | Free Full-Text | Oxidation of Organic Compounds with Peroxides Catalyzed by Polynuclear Metal Compounds
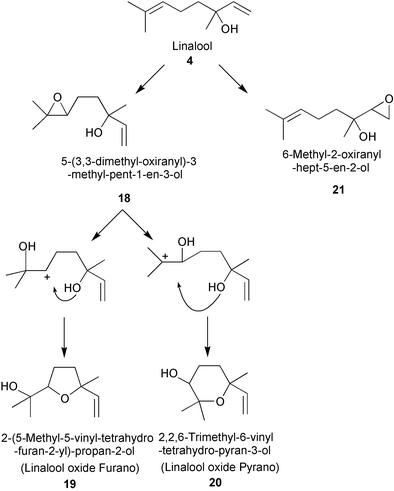
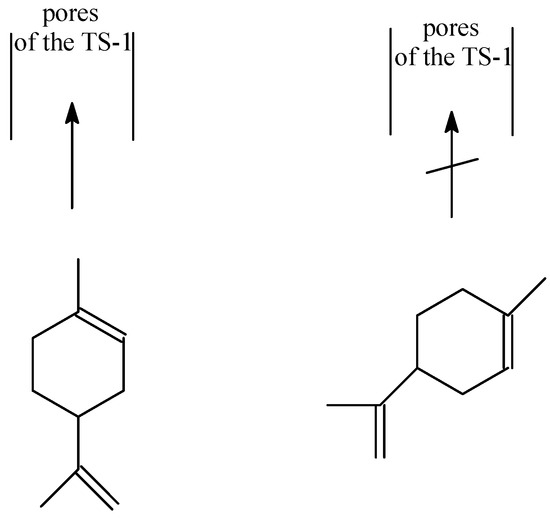
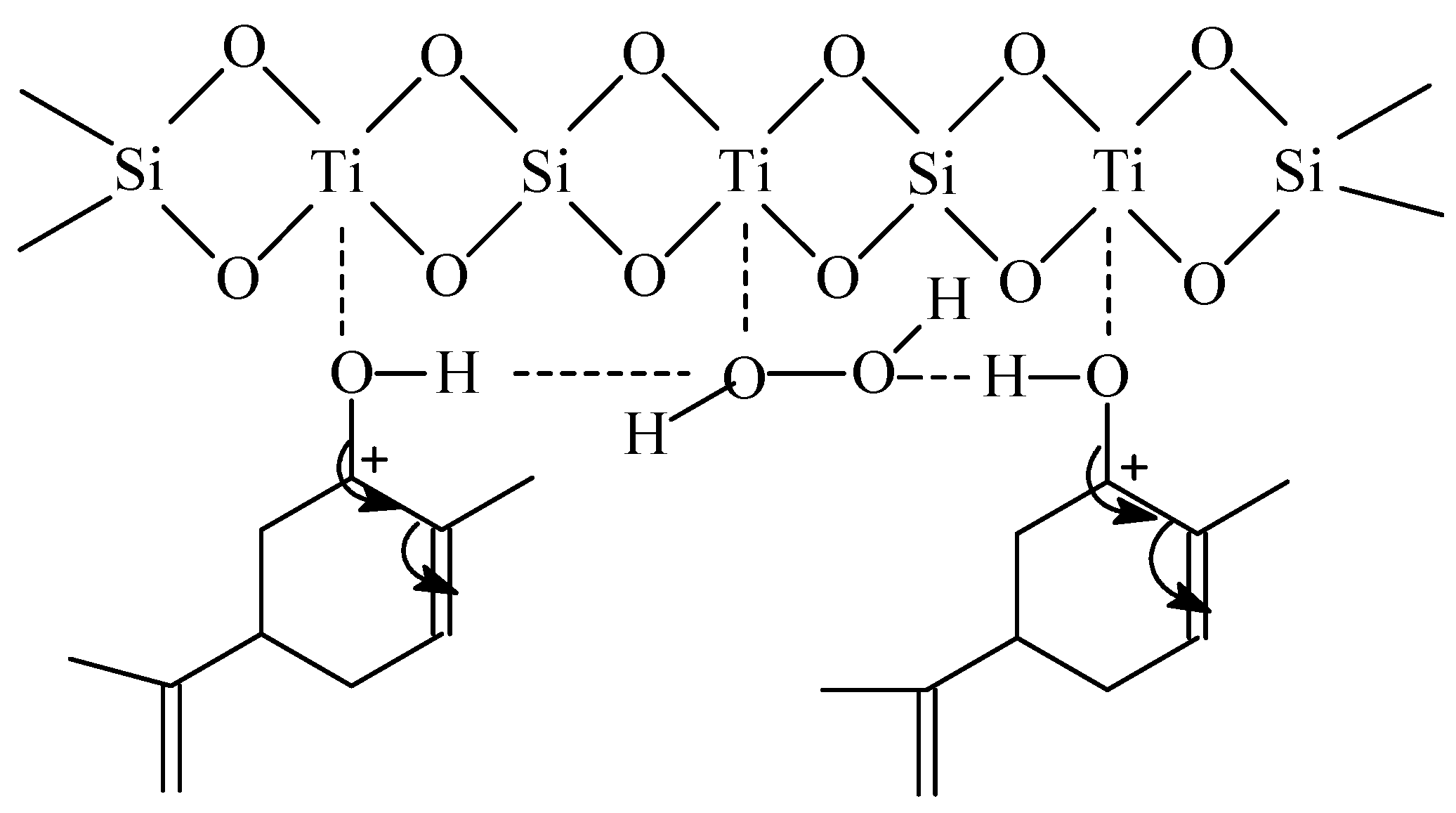
Oxidation of α-hydroxy containing monoterpenes using titanium silicate catalysts : comments on regioselectivity and the role of acidity - Journal of the Chemical Society, Perkin Transactions 2 (RSC Publishing) DOI:10.1039/B201724P

Oxidations by the system “hydrogen peroxide–dinuclear manganese(IV) complex–carboxylic acid”: Part 5. Epoxidation of olefins including natural terpenes - ScienceDirect

Typical reaction scheme for the epoxidation of limonene with hydrogen... | Download Scientific Diagram

Intensification of oxidation and epoxidation reactions—Microwave vs. conventional heating - ScienceDirect